
These highlights do not include all the information needed to use ACTHAR® GEL safely and effectively. See full prescribing information for ACTHAR GEL. ACTHAR GEL (repository corticotropin injection), for intramuscular or subcutaneous

Description of Registry Cohort. Figure 2: Percent of patients on Acthar... | Download Scientific Diagram
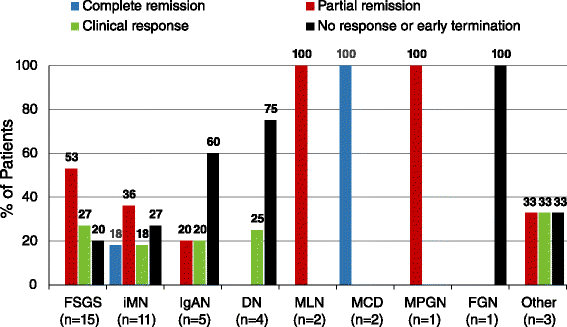
Acthar gel in the treatment of nephrotic syndrome: a multicenter retrospective case series | BMC Nephrology | Full Text

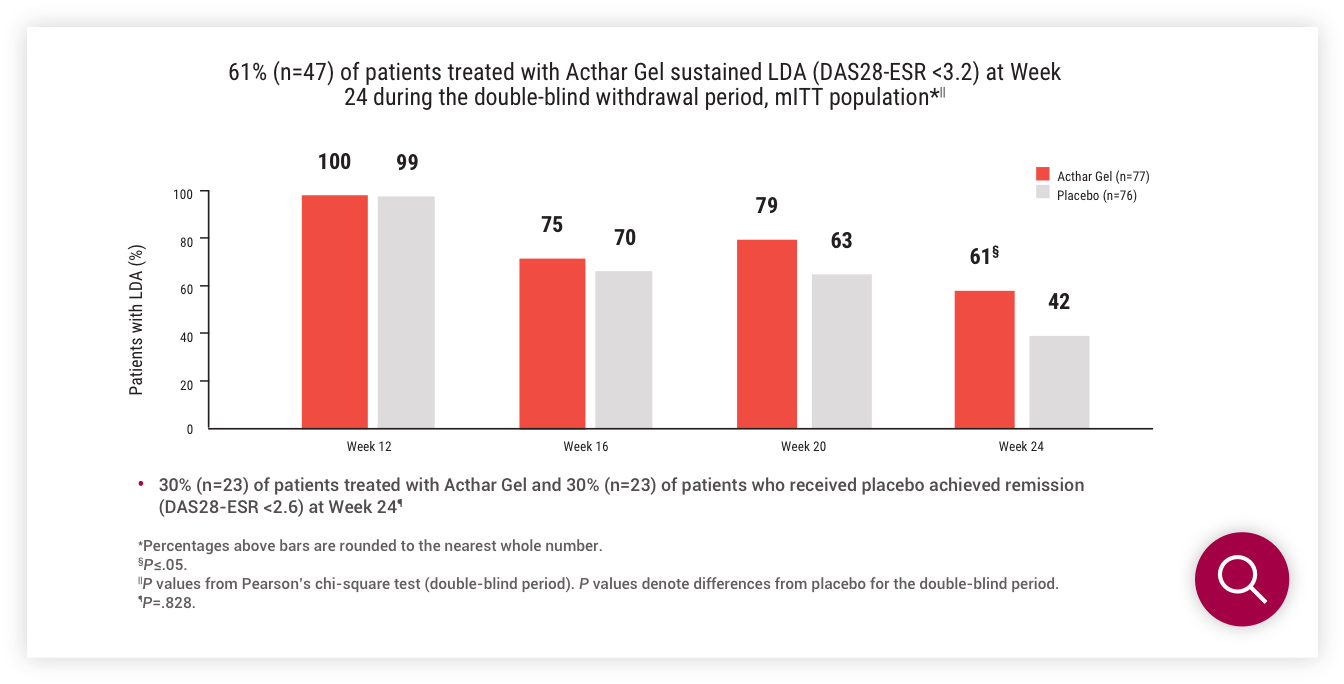





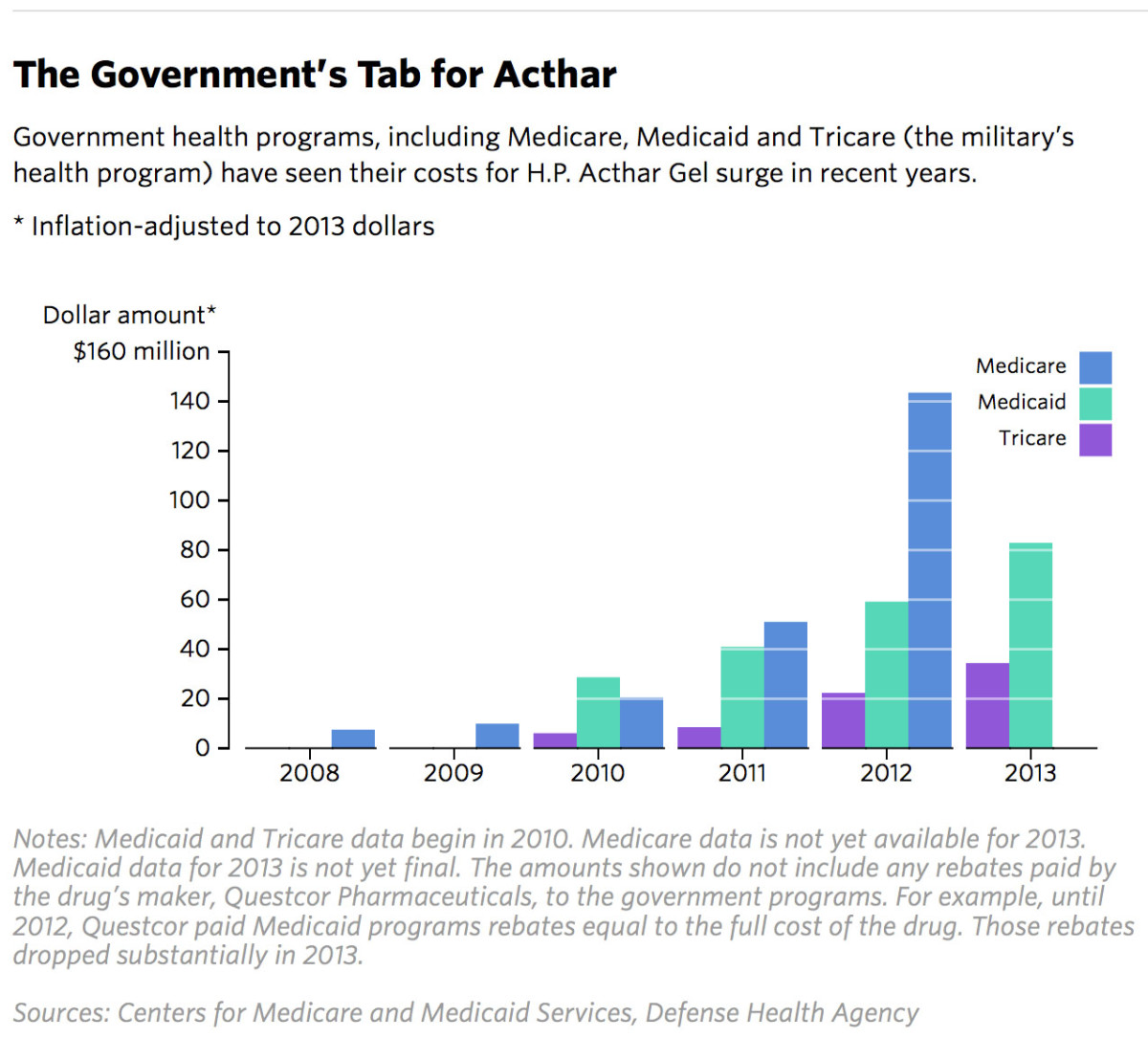










/cloudfront-us-east-1.images.arcpublishing.com/gray/W2EVDH77SVLWTNGIJIH57M6TRA.jpg)
